How Scientists Are Defeating the Citrus Psyllid with Genetics

Take an extreme close-up look at HLB’s vector, the citrus psyllid.
Photo by Jeffrey W. Lotz
There were plenty of topics covered at this year’s Florida Citrus Growers’ Institute. There was an emphasis on what can be done now to manage HLB through psyllid control and promoting tree and root health. But one presentation featured a more long-term solution surrounding involving RNAi and genetics. While still a ways off, this research could represent a more viable fix to HLB.
Bill Dawson, Eminent Scholar of Plant Pathology with UF/IFAS, presented research on the use of RNAi genetics and how it might one day be used to manage HLB or the psyllid. He told attendees that RNAi is a major defense mechanism found in plants and animals. RNAi science is still fairly new, so researchers are only more recently studying how it might be employed to fight HLB.
“RNAi is in fact a major defense mechanism against viruses, so once you understand [how it works] in plants, animals, and insects, you can learn how to change it for your own goals,” Dawson said. “So, we are using the citrus tristeza virus (CTV) to express genes in plants.”
Dawson said there are two basic ways to express genes in plants; one way is through transgenics in which a gene from another species is introduced into the plant; another way is to use a virus-based vector.
“The major advantage of the viral vector is that it is faster [than transgenics],” Dawson said. “It is not better because it is not permanent. Eventually, the plant loses the gene you put in, but you can get it into the trees much faster.”
The process of using CTV as a viral vector was developed by UF/IFAS. The patent for the technology was sold to Southern Gardens Citrus. The company is using the technology to deliver a spinach gene into trees, which would defend against HLB. The company is currently working through the regulatory process to hopefully bring the technology to groves one day soon.
Meanwhile, about 10 laboratories across the country are working on ways to deliver RNAi into citrus. When double-stranded RNA is introduced into plants, there is a response in which the RNA is chewed up into smaller RNA, which becomes a guide to go look for the target gene sequences, and it essentially chews up those sequences.
Dawson said there also has been a lot of emphasis placed on finding antimicrobial peptides that would disrupt the HLB bacteria. While there have been some identified that have activity and give plants some degree of tolerance to HLB, the search continues for even better agents.
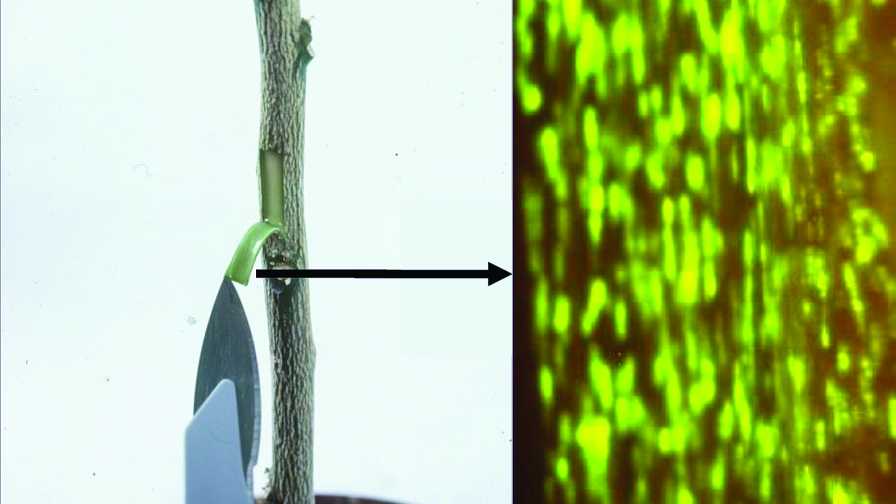
A viral vector was used to introduce a gene from a jellyfish into a citrus plant, which causes the phloem to glow.
Photos courtesy of UF/IFAS
Guns and Ammo
Dawson said you could consider the CTV viral vector the gun and antimicrobial peptides and RNAi mechanisms the bullets.
“The idea is to express antimicrobial peptides into plants as Southern Gardens is trying to do,” he said. “But, at the same time, we would like to add small RNAi sequences that would target the psyllid. So, we could try to control bacteria and the vector at the same time.”
The process of engineering CTV to become a viral vector can provide some pretty cool results. Researchers have inserted a gene from a green jellyfish into citrus. You can see where the virus has gone in the plant and introduced the jelly fish gene by peeling back the bark in a dark room and shining it with a UV light. You can see the infected cells glow.
The CTV vector is phloem-limited, meaning it only travels in the phloem of the trunk and veins in leaves. This works with HLB because it targets the phloem.
“We are very interested in this because the virus resides in the place as CLas [HLB bacteria],” Dawson said. “If we can get the virus to introduce something that infers with the HLB bacterium, we might have a chance to control the disease. The remarkable thing is the virus is pretty stable. We made some trees in 2003 with the glowing phloem. Most of those trees still have the green florescent protein. Some have lost it after a period of time; but in general, the genes will keep working for several years.”
This genetic trick allows scientist to manipulate citrus plants. If something is found that will work against the psyllid or HLB, it is easy to introduce and propagate via the viral vector.
“You can use infected leaf pieces or bark and graft it onto an another tree and introduce the virus into the tree, along with whatever extra gene you put in with the virus.” Dawson said. “The gene will continue to express in the tree.”
Through grafting, it would be easy to pre-infect budwood trees. Those buds grafted onto rootstocks in the nursery would pass along the virus and the extra genes or bullets desired to be delivered into the trees.
“What we are most interested in is if we can find a really good bullet protein or RNAi that will affect CLas, with the viral vector, we could put it in existing trees in the field,” Dawson said. “You could do therapy.”
Finding the Right Bullet
RNAi can be used to suppress gene functions. Researchers are seeking to use RNAi to knock out a gene or enzyme the psyllid really needs to function or survive.
“If the psyllid consumes those small RNAs into their gut, the psyllid has the same machinery, so those small RNAs will be incorporated into the psyllid’s machinery,” Dawson said. “The messenger RNA goes looking for the [target] gene and knocks it out.”
That would be the end of the psyllid. Researchers from UF/IFAS and other labs are screening RNAi that would achieve this result. Some sequences seem to be working pretty well, while others do not.
“RNAi has only been discovered in the past 20 years, but it has tremendous potential as an insecticide because it is based totally on a sequence of RNA,” Dawson said. “In theory, we can make a toxic sequence to any insect that will be specific only to that pest.”










